You’ve decided who is going to formulate your dream product. The next step is to ensure that you can properly...

Ocean safety and sunscreen ingredients
Think you know all about reef safety, coral bleaching and reef safe products? You might be surprised. Click here to read our overview blog – Reef safe products – crisis or con?
What’s it all about?
- Where contamination is unavoidable, use chemicals that are ecologically safe to minimise harm
- Non ecologically safe sunscreen actives include all forms of Zinc Oxide (which are incorrectly believed to be reef safe), along with Octocrylene, Octyl Salicylate and Benzophenone-3 (Oxybenzone)
- Protective clothing and minimising sunscreen use can help minimise the impact on oceans
- Highly water resistant sunscreens help minimise unwanted contamination of the ocean environment
Zinc Oxide – not all it’s cracked up to be?
In this blog it’s discussed where the claims of ‘reef safe’ products and chemicals originated; a US-based environmental laboratory undertaking experimental research that aimed to directly link a sunscreen active to coral bleaching.
Contrary to the laboratories suggestion that ‘non nano’ Zinc Oxide is a non marine pollutant, and by association, non coral bleaching (somehow excluding nanoparticles), there is research that suggests uncoated Zinc Oxide can contribute to coral bleaching and the European Chemicals Agency (ECHA) aquatic hazard classification categorised Zinc Oxide as being very toxic to marine life with long lasting effects.
Primary vs secondary particles sizing

The laboratory implies that ‘non nano’ Zinc Oxide(i.e. larger than 100 nm in size) is miraculously safe as compared to ‘nano’ Zinc Oxide (i.e. less than 100 nm in size), as one may expect, safety and toxicity associated with ‘small’ and ‘smaller’ is unlikely to be that black and white (101nm is ok?).
The vast majority of sunscreen grade Zinc Oxide have what we refer to as ‘primary particles’ that are nano in size, which is what makes them highly effective sunscreens. It is these primary particles that then form tightly held, larger aggregates which are referred to as the ‘secondary particle’ that by definition makes them non nano (despite being nano).
Beyond ‘reef safe’ to ‘ocean safe’
Over the past decade, consumers have increased their focus on carbon footprints, global warming and harm minimisation for future generations.
Whilst the concern that sunscreens are impacting reefs is likely to be largely unwarranted, in the context of global warming and increasing ocean temperatures, we should continue to minimise our impact on oceans which of course affects other marine life including fish, crustaceans, molluscs etc, particularly in areas that have a rich ecosystem, which is often around reefs.
How do we protect ourselves and the oceans?
Whilst we must care for the environment and do everything we can to minimise unnecessary damage, we need to also protect ourselves. The harm sunscreens may cause to coral reefs is not well defined, in contrast, the harm not wearing sunscreen causes to humans is undeniable and we must find a balance.
Some suggestions include:
- Swim in a pool and not in the ocean if possible. Whilst pool water will be contaminated and eventually end up in the sewer system, the treatment process can be expected to remove contaminants with effluent kept away from beaches and reefs
- If swimming in beaches, pick ones away from reefs and other rich ecosystems
- Wear protective clothing (high UPF long sleeve rash vests, hats etc) to minimise the amount of skin requiring sunscreen
- Do not automatically go for ‘mineral’, ‘natural’ and ‘non-nano’ sunscreens which are often perceived as safest
- Use an eco-friendly sunscreen, avoid anything with Zinc Oxide, Octocrylene, Benzophenone-3 and Octyl Salicylate
- Ensure the sunscreen has the maximum water resistance possible, an Australian-made 4hr water resistant sunscreen is always preferable over an 80 minute resistance
What is an eco-friendly sunscreen?
Although the majority of sunscreens are perfectly fine for everyday use in and around a pool, when selecting sunscreens to use in lakes, rivers and beaches, we should avoid sunscreens that use active ingredients associated with eco toxicity where possible, using the ECHA classifications as a guide.
ECHA eco toxicity classifications of sunscreen active ingredients
HIGH RISK
- Zinc Oxide
- Octocrylene
- Octyl Salicylate
- Benzophenone-3 (Oxybenzone)
LOW RISK
- Octyl Triazone
- Octyl Methoxycinnamate (Octinoxate)
- 4-Methylbenzylidene Camphor
Instead, focus on ingredients that haven’t been associated with causing toxicity or have otherwise negligible levels of risk.
POSSIBLE RISK
- Methylene Bis-Benzotriazolyl Tetramethylbutylphenol
- Diethylamino Hydroxybenzoyl Hexyl Benzoate
NEGLIGIBLE RISK
- Titanium dioxide
- Phenylbenzimidazole Sulfonic Acid
- Homosalate
- Avobenzone
- Bis-Ethylhexyloxyphenol Methoxyphenyl Triazine
- Tris Biphenyl Triazine
The EcoSun Pass
EcoSun is a proprietary system developed by global leader in sunscreen chemical development and the world’s largest chemical producer, BASF. The EcoSun system quantifies the environmental safety of sunscreen actives and their usage concentrations based on a variety of factors including:
- Acute aquatic toxicity
- Chronic aquatic toxicity
- Biodegradability
- Bioaccumulation
- Endocrine suspicion
- Terrestrial toxicity
- Sediment toxicity
To achieve EcoSun Pass certification, BASF set a target score of >200 however, the algorithm and combination of the above factors is proprietary and therefore unknown.
An example of how the EcoSun system works; at a 10% dosage of a single sunscreen chemical, the ‘high risk’ ingredients score poorly; Zinc Oxide a paltry ‘37’, Octocrylene, Octyl Salicylate ‘0’ and Benzophenone ‘77’. In contrast, the negligible risk ingredients score far better; Bemotrizinol ‘302’, Titanium Dioxide ‘148’ (still not as good as some modern synthetic chemicals) and Tris-Biphenyl Triazine ‘189’.
We did find a minor flaw in the algorithm logic, where we would assume using less of any chemical ingredient would be better for the environment and result in a higher score, or at a minimum, have the same score regardless of dose which enables a EcoSun rating against active concentration. Unfortunately, in some cases the system appears to encourage higher usage levels to achieve higher ratings, which is counterintuitive, as the best scores should be as the dosage approaches zero and less actives enter the environment.
As an example a 50/50 blend of Bemotrizinol and Tris-Biphenyl Triazine:
5% – 268
10% – 285
15% – 300
20% – 312
Admittedly the scores are quite good and all exceed the 200 required for certification however this is not factoring in the resulting SPF. In combination with other sunscreen chemicals, it is perhaps pushing BASF proprietary chemicals and less economical active ingredients.
Oddly, this flaw was corrected when using another active (Avobenzone) and less erratic when looking at others individually, all that aside, this is a great tool.
Avobenzone:
2.5% – 149
5.0% – 98
7.5% – 69
10% – 53
Interestingly, it seems individual ingredients that scored zero, including Octocrylene and Octyl Salicylate, would cause an otherwise favourable combination of ingredients to immediately be scored zero at any combination.
Combining EcoSun value with the BASF sunscreen simulator (which enables in silico estimates of sunscreen performance), a high-performing sunscreen capable of an EcoSun Pass of 238 is feasible using the following sunscreen chemicals:
Titanium Dioxide, Tris Biphenyl Triazine, Phenylbenzimidazole Sulfonic Acid, Butyl Methoxydibenzoylmethane & Bis-Ethylhexyloxyphenol Methoxyphenyl Triazine
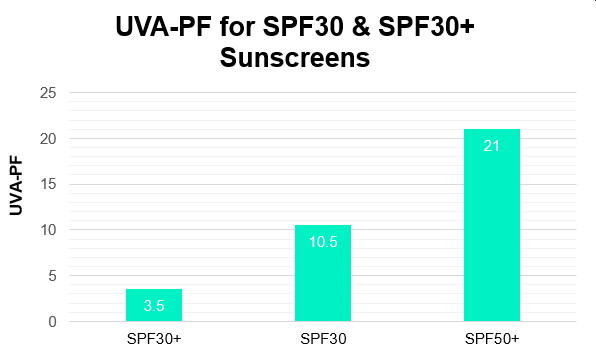
The above combination was estimated to achieve an SPF of 62.5 and a UVA-PF of 21.4
An all-mineral formulation based on either Zinc Oxide, Titanium Dioxide or a combination of both are not capable of achieving broad spectrum SPF50+ and an EcoSun value of >200.
Maximise the water resistance and minimise the contamination
Having an entirely waterproof sunscreen would be a solution to the concern of sunscreen chemicals entering waterways. However whilst an entirely waterproof sunscreen may not be practical, some sunscreens are far more resistant to wash off than others, and the ‘water resistance’ claims on packaging only go so far as to communicating how resistant they are.
Australian water resistant sunscreens are on balance more water resistant than their overseas counterparts, as a consequence of how the test results are interpreted to make the claim, but also as a result of higher time periods (80 minutes vs 4 hours).
Sunscreens that make water resistance claims in Australia are required to maintain the claimed amount of SPF after immersion in water. So if claiming SPF50 and 4hr water resistance, the sunscreen must still be SPF50 after 4 hrs. In contrast, a US or EU SPF50 sunscreen, which can only claim 80 minutes water resistance (1/3rd of what an Australian product can), is permitted to be SPF 25 after 80 minutes.
What this means in simplistic terms is that half of the sunscreen is allowed to wash off after 80 minutes, potentially it has all washed off after 4 hours, whereas the Australian sunscreen has not washed off at all after that same time period.
Worst-case allowable SPF claims and water resistance

This is a fairly extreme example and there are likely to be European sunscreens that are extraordinarily water resistant by chance rather than design.
Unfortunately, there is currently no measure of water resistance indicating ‘how much will end up in the ocean’, something that could become a feature of future sunscreens or a proven claim relating to ocean safety.
How do I draft a product formulation brief?
How do I formulate a product?
So, you have a great idea for a new product, but you aren’t sure what’s next? There are a few steps between a wonderful idea and uploading products to your website or being stocked at your favourite retailer
Safe or sorry: the perils of preservative free skincare
Products making ‘preservative free’ claims are becoming increasingly common. While there are legitimate and safe ways...